Post-Herpetic Neuralgia: When Shingles Pain Doesn't Go Away
When the shingles rash heals but the pain remains, or worsens, this is not a failure of healing. It is a sign that the nerve itself has been injured.
Post-herpetic neuralgia is one of the most painful and most misunderstood complications of shingles. The skin looks normal. The infection is over. Yet for many people, the pain continues, sometimes for months, sometimes for years.
What You Need to Know
PHN affects an estimated 5 to 15 per cent of people who have shingles, rising sharply in adults over 80PHN affects an estimated 5 to 15 per cent of people who have shingles, rising sharply in adults over 80.
Post-herpetic neuralgia (PHN) is persistent nerve pain that continues after a shingles episode, typically defined as pain lasting more than three months from rash onset.
The rash clearing does not mean the nerve has recovered. The varicella-zoster virus can cause lasting injury to the spinal relay nerve (dorsal root ganglion) and surrounding nerve fibres.
Burning pain, electric sensations, and pain from light touch or clothing (allodynia) are signs of nerve sensitisation, not normal post-shingles healing.
Gabapentinoids like gabapentin and pregabalin can help some patients but do not fully resolve PHN in many cases. Targeted interventional options exist for those who do not respond adequately.
Earlier specialist assessment is associated with better outcomes. The longer PHN persists, the more established the pain pathway can become and harder to treat.
When the shingles rash heals but the pain remains, or worsens, this is not a failure of healing. It is a sign that the nerve itself has been injured.
Post-herpetic neuralgia is one of the most painful and most misunderstood complications of shingles. The skin looks normal. The infection is over. Yet for many people, the pain continues, sometimes for months, sometimes for years.
This happens because shingles does not just irritate the surface. The varicella-zoster virus travels along nerve fibres and can cause lasting damage to the nerve itself. Once that injury is established, the pain is no longer a symptom of infection. It is the result of a damaged and sensitised nerve continuing to fire abnormal pain signals.
Understanding what is driving your pain is the first step toward managing it. If you are unsure whether what you are experiencing is post-herpetic neuralgia, a specialist assessment can provide clarity.
What is post-herpetic neuralgia?
Post-herpetic neuralgia is defined as persistent pain in the area affected by a shingles episode that continues for three months or more after the rash first appeared. Some clinical definitions use four weeks from rash healing as the threshold. Whichever definition is used, the underlying issue is the same: the nerve has been damaged, and the pain has outlasted the infection.
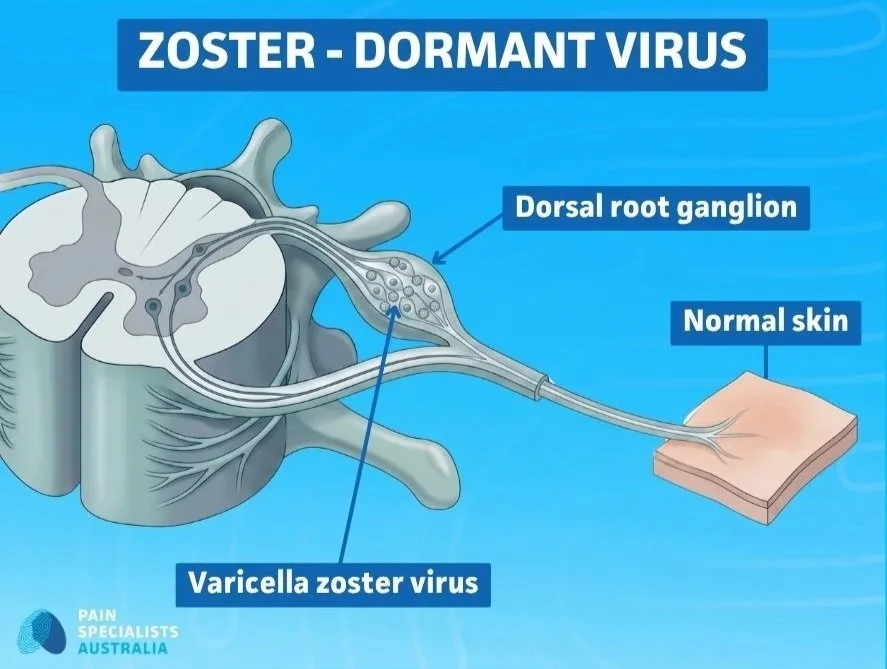
Shingles itself is caused by the reactivation of the varicella-zoster virus; the same virus responsible for chickenpox. After a chickenpox episode, the virus lies dormant in the nerve roots.
The varicella-zoster virus lies dormant in the dorsal root ganglion until reactivation.
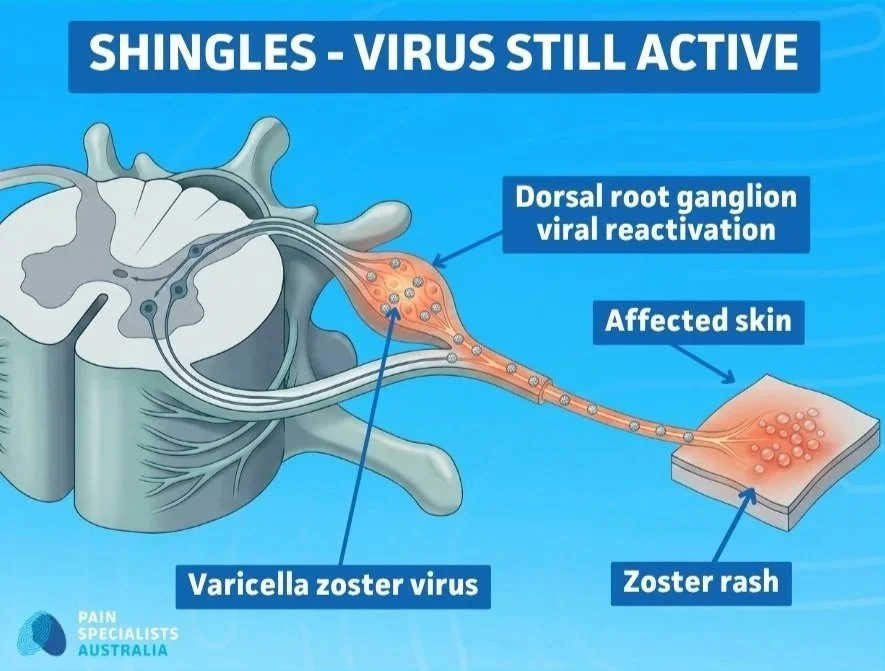
In some people, particularly those with reduced immunity, it reactivates decades later as shingles. The resulting rash is painful and typically follows a dermatomal pattern, a band or stripe of blistering skin on one side of the body that corresponds to the distribution of a single nerve.
For most people, the acute pain of shingles resolves as the rash heals. For others, the nerve does not fully recover, and the pain persists.
How PHN differs from the pain of an acute shingles episode
Acute shingles pain is painful and distressing, but it has a clear cause: active viral inflammation along the nerve. As the immune system controls the virus and the rash resolves, pain typically improves.
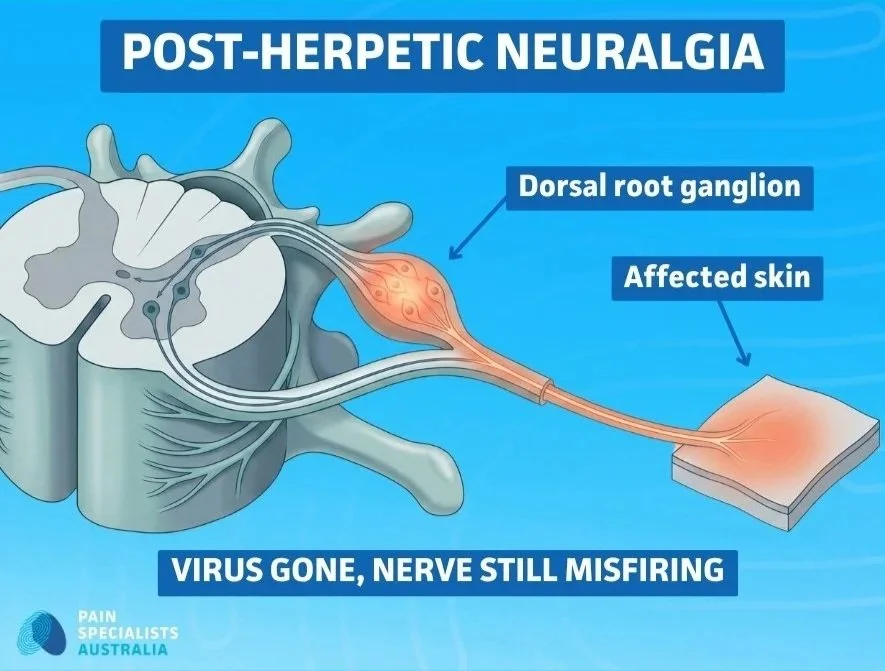
Post-herpetic neuralgia is a different condition. The virus is no longer active, but the nerve has been structurally damaged. This means treatment that targets infection will have no effect on PHN. The target must be the nerve, the pain signalling pathway, or both.
This distinction matters because some patients are told, or come to believe, that their ongoing pain means the shingles is still active. In most cases, it is not. The nerve has been injured, and the pain signals it is generating are not a sign of ongoing infection.
Who is most at risk of developing PHN after shingles?
PHN does not affect everyone who has shingles. Risk is significantly higher in:
Adults over 60, with risk rising sharply in those over 80
People with severe or widespread shingles rash at the time of the initial episode
People who experienced intense pain during the acute shingles phase
Those with weakened immunity due to medical conditions or medications like steroids
People in whom antiviral treatment was delayed or not given
Those with shingles affecting the face or head (ophthalmic distribution)
People who have not had the shingles vaccine
Incidence estimates vary but PHN is believed to affect somewhere between 5 and 15 per cent of people who experience shingles, with rates much higher in older age groups.
Why shingles pain persists - what is happening to the nerve
Understanding why PHN persists requires understanding what the varicella-zoster virus actually does to nerve tissue. This is not a simple story of inflammation that heals. It is a story of nerve injury that can alter pain signalling in lasting ways.
Nerve injury, not ongoing infection
When the varicella-zoster virus reactivates, it travels along the sensory nerve from the dorsal root ganglion toward the skin. As it does so, it causes direct inflammatory damage to the nerve fibres. In many people, this damage is short-lasting and the nerve fully recovers. In others, the injury is sufficient to alter how the nerve functions on a lasting basis.
After the virus is cleared, these damaged nerve fibres may begin to generate spontaneous electrical signals, firing abnormally without any trigger. They may also become more sensitive to stimuli that would not normally cause pain, such as light touch, temperature change, or even air movement across the skin.
The result is pain that feels very real, is very real, but has no active infection causing it.
The virus is gone. The nerve injury remains.
The dorsal root ganglion - the structure the virus damages
The dorsal root ganglion (DRG) is a cluster of sensory nerve cell bodies located just outside the spinal cord, one at each spinal level on each side of the body. It is the key relay point for sensory information travelling from the body into the central nervous system.
The varicella-zoster virus has a particular affinity for the DRG. This is where the virus lies dormant between episodes, and it is the site of significant inflammation and injury when the virus reactivates. DRG cell bodies can be directly damaged during a shingles episode, and this damage can alter how the entire nerve behaves long after the acute infection has resolved.
During a shingles episode, the varicella-zoster virus reactivates in the dorsal root ganglion and travels along the nerve to the skin - causing the characteristic blistering rash in a dermatomal pattern.
Post-herpetic neuralgia follows the dermatomal distribution of the nerve damaged during the shingles episode.
This is clinically important because treatments that target the DRG directly, rather than broadly suppressing nerve activity across the whole system, may offer more effective relief for some patients with PHN.
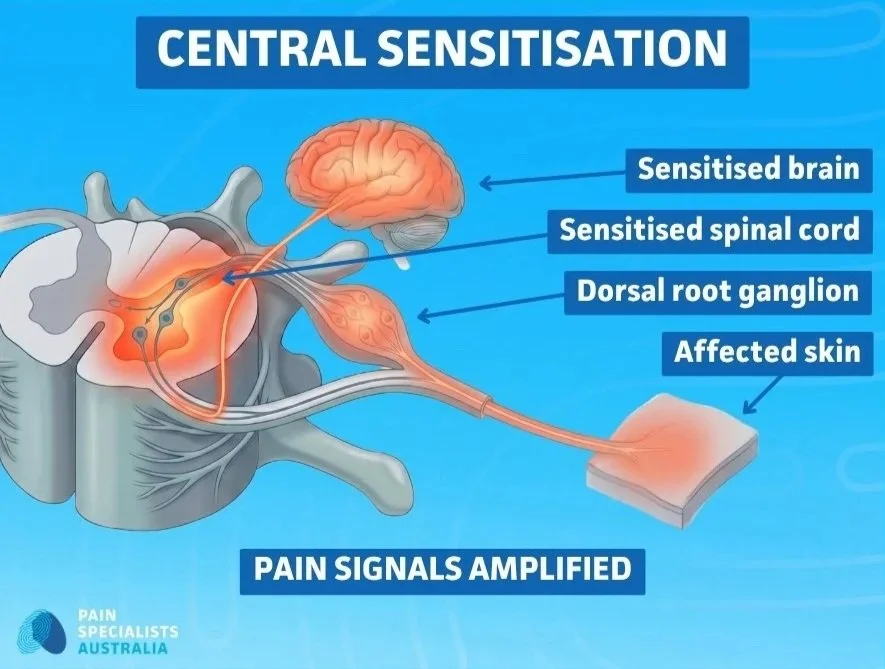
Central sensitisation - why PHN can become harder to treat over time
When peripheral nerve fibres fire abnormally over a sustained period, the central nervous system, the spinal cord and brain, can begin to amplify those signals. This process is called central sensitisation.
In practical terms, this means that the pain processing system itself becomes more reactive. The threshold for pain is lowered. Stimuli that should not cause pain begin to register as painful. The volume of the pain signal is turned up beyond what the peripheral injury alone would produce.
Central sensitisation is not a psychological phenomenon. It is a biological process with measurable changes in spinal cord and brain function. However, it does mean that PHN which has been present for a prolonged period can become more complex and more resistant to treatment than PHN addressed early. This is one of the strongest arguments for seeking specialist assessment sooner rather than later.
In PHN, prolonged nerve firing can sensitise the spinal cord and brain, amplifying pain signals beyond the original site of injury.
What post-herpetic neuralgia feels like
PHN presents differently in different people, but certain patterns are common and clinically recognisable. These patterns reflect the underlying nerve injury rather than ongoing skin or tissue damage.
Allodynia - when light touch, clothing, or temperature cause pain
One of the most debilitating features of PHN is allodynia, a condition in which normally non-painful stimuli cause significant pain. In the context of PHN, this often means:
Clothing resting against the affected skin causes intense burning or stinging
Bedsheets touching the area at night make sleep difficult or impossible
Water from a shower or bath is acutely painful in the affected region
A change in temperature or air movement across the skin triggers pain
Any light pressure on the skin, including examination, produces a disproportionate pain response
Allodynia is one of the clearest signs of nerve sensitisation. It indicates that the nerve is no longer calibrating pain signals accurately. It is amplifying normal sensory input as if it were harmful. Allodynia is a common feature of nerve pain in general.
Clinically, the presence of allodynia alongside the history of a shingles episode in that distribution is a strong indicator that the pain is neuropathic and related to the original nerve injury.
Pain patterns that suggest nerve involvement rather than normal healing
PHN pain is typically described in ways that differ meaningfully from musculoskeletal or inflammatory pain. Common complaints include:
Burning: a constant deep heat or surface heat in the skin
Electric shocks: sudden sharp jolts of pain that come without warning
Stabbing or shooting sensations
Raw or tender skin, as if sunburned, even in the absence of any visible change
A band or stripe of pain that follows the original rash distribution
Numbness or altered sensation alongside the pain
When patients describe a combination of burning, sensitivity to touch, and shooting sensations in the area of a previous shingles rash, particularly when the skin now looks normal, this is a recognisable pattern that warrants thorough neuropathic pain assessment rather than reassurance that the shingles is over.
If your shingles pain has persisted beyond what you expected, or has worsened since the rash resolved, a specialist assessment can help identify what is happening and what options may be available. You do not need to wait until the pain is unbearable to seek a clearer answer.
Why some patients need more than medication
First-line medications for PHN, primarily gabapentin, pregabalin, and tricyclic antidepressants, are effective for some patients. Many patients experience meaningful improvement with these treatments. But a significant proportion do not achieve adequate relief, and some find that side effects limit how much benefit they can practically obtain.
What gabapentin and pregabalin do - and why they help some patients more than others
Gabapentin and pregabalin work by reducing the excitability of nerve fibres throughout the nervous system. They act on calcium channels in nerve cells and can dampen abnormal nerve firing across a wide network of nerves.
This systemic approach is useful when the pain is diffuse, when sensitisation has spread broadly. But in PHN, particularly in the earlier stages, the primary driver is often located at or near a specific dorsal root ganglion that was damaged by the virus. A systemic medication that acts broadly across the whole nervous system may not deliver sufficient pain reducation at the specific site of injury without requiring higher doses that produce significant side effects. Sedation, cognitive dulling, balance problems, and fatigue are among the most common side effects.
This is one of the reasons why targeted interventional approaches, delivered directly to the relevant DRG, could offer relief for patients who have not responded adequately to oral medications alone.
Why timing matters - the case for early specialist assessment
Evidence consistently supports the principle that earlier treatment of PHN is associated with better outcomes. There are two main reasons for this.
First, before central sensitisation becomes fully established, the primary driver of pain is more likely to be peripheral, located at the damaged DRG. This makes interventions targeting the DRG more likely to be effective. Once central sensitisation has occurred, treatment becomes more complex and outcomes less predictable.
Second, prolonged severe pain has functional consequences. Sleep disruption, fatigue, reduced mobility, social withdrawal, and the psychological burden of uncontrolled pain all compound over time. Treating the pain pathway earlier reduces the impact of these other broader effects.
Patients who have been managing PHN for months on the same treatment regimen without adequate relief, or who are experiencing significant side effects from medication, should be assessed by a specialist pain physician before assuming that medication represents the limit of what can be done.
Shingles Pain That Won't Stop: The Targeted Treatment Most Patients Haven't Heard About
This video provides general information only and is not a substitute for individual medical advice. Outcomes and treatment suitability vary.
Accessing this content does not establish a doctor-patient relationship. Any case studies mentioned are hypothetical.
Treatment options for post-herpetic neuralgia
There is no single treatment that resolves PHN in every patient. The most appropriate treatment depends on the duration of the pain, the predominant mechanism driving it, the area of the body affected, and how the patient has responded to prior management. At Pain Specialists Australia, treatment is preceded by thorough clinical assessment and diagnostic reasoning, identifying what is likely driving the pain before deciding how to treat it.
Medications
A range of medications have demonstrated efficacy in PHN and remain part of the management pathway for most patients.
Anticonvulsants (gabapentinoids): Gabapentin and pregabalin are commonly used as first-line agents. They reduce nerve excitability and can provide meaningful relief in patients with PHN, particularly those with prominent burning and allodynia. Response varies between individuals, and dose titration is often required.
Tricyclic antidepressants: Amitriptyline and nortriptyline are used in low doses for their neuropathic pain modulating properties rather than their antidepressant effect. They can be effective but are limited in older patients due to side effects including sedation and cardiac effects.
Topical agents: Lignocaine patches and topical capsaicin (including the high-concentration 8% capsaicin patch) can provide localised relief with reduced systemic side effects. These are particularly relevant where allodynia is a prominent feature and where the affected area is accessible and not too extensive.
Palmitoylethanolamide (PEA): A supplement anti-inflammatory with neuromodulatory properties, PEA has been studied in neuropathic pain including PHN. While evidence continues to evolve, it is considered a reasonable add on option in the management of PHN, particularly where tolerability of other agents is a concern.
Opioids: Opioids are occasionally used in carefully selected patients for refractory PHN when other approaches have failed. The decision to use opioids in this context requires careful clinical assessment, monitoring, and alignment with current prescribing guidance. Sometimes opioids can cause Opioid-Induced-Hyperalgesia (OIH) (read more about this in our blog article discussing OIH here.
Medication selection should be individualised. Combination approaches are often necessary. The goal is to achieve meaningful functional improvement while maintaining tolerability.
Pulsed radiofrequency of the dorsal root ganglion
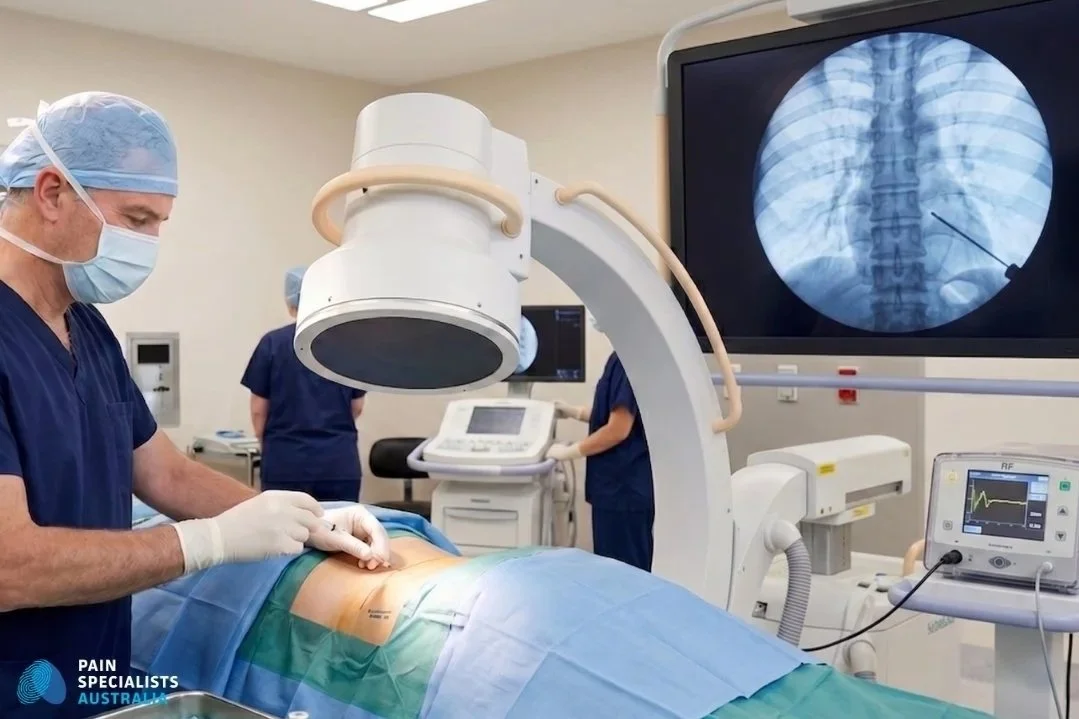
Pulsed radiofrequency (PRF) of the dorsal root ganglion is one of the most evidence-supported interventional treatments for PHN and represents a significant option for patients who have not responded adequately to medication.
PRF is performed under fluoroscopic (X-ray) guidance by a specialist pain physician. A needle is positioned next to the relevant DRG, the nerve cluster at the spinal level corresponding to the area affected by PHN. A pulsed, non-destructive radiofrequency current is then applied to the ganglion.
Unlike conventional radiofrequency ablation, pulsed radiofrequency does not destroy nerve tissue. It is thought to modulate how the DRG processes and transmits pain signals, reducing the abnormal firing that drives PHN pain, without permanent structural change. This makes it a suitable option even when the affected nerve is sensory and long-term preservation of sensation is important.
The procedure is performed as a day procedure. Most patients return home the same day.
Pulsed radiofrequency is generally considered a low-risk procedure, though as with any interventional treatment, risks include temporary discomfort at the injection site, bruising, and in rare cases infection, nerve irritation, or nerve damage.
Pulsed radiofrequency of the dorsal root ganglion is performed under fluoroscopic guidance by a specialist pain physician.
Multiple randomised controlled trials have demonstrated meaningful reduction in PHN pain scores following PRF of the DRG, including in patients with significant allodynia and in patients who have been inadequately controlled on oral medications. Evidence supports its use both in thoracic-distribution PHN (the most common) and in cervical or lumbar-distribution cases. The evidence is moderate.
For patients whose PHN is driven primarily by ongoing DRG dysfunction, particularly those with a duration of less than two years and identifiable segmental distribution, PRF of the DRG offers a targeted, and reversible option that should be discussed as part of the treatment pathway when first-line management is insufficient.
Nerve blocks - including stellate ganglion block for facial and cervical PHN
Sympathetically mediated components may contribute to PHN pain in some patients, particularly those with ophthalmic or cervical distribution (shingles affecting the face, head, or upper chest). In these cases, nerve blocks targeting the relevant sympathetic pathways can be clinically useful.
Stellate ganglion block: The stellate ganglion is a sympathetic nerve cluster in the neck region. Blockade of the stellate ganglion can reduce sympathetically maintained pain in cervical and upper thoracic PHN, and in some cases in ophthalmic distribution PHN. It is performed under ultrasound or fluoroscopic guidance and is a recognised option in the management of head, face, and upper limb shingles-related pain.
Epidural nerve blocks: In thoracic distribution PHN, epidural injections of local anaesthetic and corticosteroid can provide diagnostic information about nerve involvement and in some cases contribute to treatment, particularly when performed in the earlier stages of the condition.
Intercostal nerve blocks: For focal thoracic PHN, intercostal nerve blocks may offer targeted relief, particularly where a specific intercostal nerve distribution is clearly identified.
The role of nerve blocks in PHN is most clearly established in the acute and subacute phases. Evidence for long-term benefit in chronic PHN is more variable, but they may play a useful role in assessment, in combination with other treatments, or in patients where the sympathetic component appears prominent.
Spinal cord stimulation for refractory post-herpetic neuralgia
For patients with severe, long-standing PHN that has not responded to medication or other interventional treatments, spinal cord stimulation (SCS) is occasionally considered as a later-line option.
SCS involves the placement of a small lead alongside the spinal cord at the level corresponding to the affected area. A low-level electrical current modulates pain signal transmission before signals reach the brain.
SCS is not a first-line treatment for PHN and is not appropriate for most patients. The current evidence base is limited to case reports and small case series. Larger, high-quality trials are lacking. This means the evidence supporting SCS specifically for PHN is of low quality, and it should only be discussed in the context of a thorough specialist assessment for carefully selected patients where other treatments have been systematically tried and have not provided adequate relief.
Where SCS is considered, the decision involves detailed discussion of the evidence limitations, realistic expectations, and the patient's individual circumstances.
Multidisciplinary and rehabilitation support
PHN that has persisted for months or years rarely has a single treatment answer. Alongside medical and interventional management, a multidisciplinary approach can be important in restoring function, managing the burden of chronic pain, and reducing the secondary impacts that accumulate with prolonged uncontrolled pain.
This may include:
Psychology and pain-focused cognitive behavioural therapy, to address the psychological burden, sleep disruption, and behavioural adaptations that often accompany chronic PHN
Physiotherapy, particularly relevant where PHN has led to altered posture, guarding, or reduced activity
Occupational therapy, where PHN is limiting work or daily activities
Sleep support, given that night pain is one of the most disruptive features of PHN
At Pain Specialists Australia, multidisciplinary care is integrated into the management pathway for complex pain presentations. This is not a consolation pathway when interventions are declined. It is part of how chronic pain should be more broadly managed.
If your shingles pain has not responded adequately to medication, or if it has persisted beyond three months, a specialist assessment can identify the mechanism and explore whether a more targeted approach may help. Interventional options may offer meaningful relief for selected patients.
Post-herpetic neuralgia affecting the face and head
PHN does not only affect the trunk. When shingles reactivates in the trigeminal nerve distribution, particularly the ophthalmic division which supplies the forehead, scalp, and eye area, the consequences can be among the most severe and most difficult to treat of any PHN presentation.
Herpes zoster ophthalmicus (shingles affecting the eye region and forehead) carries a higher risk of PHN than many other distributions, and the pain can involve the forehead, scalp, eye, and surrounding face.
If shingles affects the eye or the skin around the eye, this requires urgent assessment by an ophthalmologist, as ophthalmic zoster can threaten vision and is considered a medical emergency.
Some patients also experience involvement of the ear, face, mouth (Ramsay Hunt syndrome) with ear (vestibular) and facial nerve consequences.
Ophthalmic and trigeminal distribution PHN - assessment and treatment
Facial and cranial PHN warrants specialist assessment that takes into account both the anatomical distribution of the pain and whether there are features suggesting sympathetic involvement, central sensitisation, or superimposed neuropathic changes in the trigeminal pathways.
Treatment options relevant to facial distribution PHN include:
Topical agents (where accessible and tolerated)
Tricyclic antidepressants and gabapentinoids
Stellate ganglion block, which can be relevant in cervical and upper cranial distributions
Radiofrequency procedures targeting the relevant ganglion (including Gasserian ganglion procedures in selected cases)
Multidisciplinary support, which is often particularly important given the impact on facial function, appearance, and identity
Patients with persistent facial pain following ophthalmic or trigeminal shingles should not be assumed to have exhausted their options after medication alone. Specialist pain assessment is appropriate and often necessary.
When to see a specialist for post-herpetic neuralgia
Consider seeking a specialist pain physician assessment if any of the following apply:
Pain has persisted for three months or more following the onset of the shingles rash
Medications (gabapentin, pregabalin, amitriptyline, or others) have provided only partial relief or have not been tolerable due to side effects
The pain is significantly affecting sleep, daily activities, or quality of life
Allodynia is present: touching the skin, clothing contact, or temperature changes are causing pain
The pain appears to be worsening rather than gradually resolving
You have been told the shingles is over but the pain does not match that reassurance
PHN is affecting the face, head, or eye area
GPs and specialists can refer directly to Pain Specialists Australia via the online referral form.
Why choose Pain Specialists Australia for post-herpetic neuralgia?
Post-herpetic neuralgia is a condition that frequently falls through the gaps of standard care.
Patients are often managed in primary care with oral medications, reassured that the shingles is over, and left without a clear explanation for why the pain continues or what else can be done. When those medications do not provide adequate relief, the options are not always clearly communicated.
Pain Specialists Australia is a specialist private pain clinic based in Melbourne, with clinics in Richmond, Heidelberg, and Bayside (Sandringham). Our specialist pain physicians have extensive experience in the assessment and management of neuropathic pain conditions, including post-herpetic neuralgia across all its distributions and stages.
What we offer patients with PHN:
Thorough clinical assessment that identifies the mechanism driving your pain, not just the diagnosis, but why it is persisting and what may help.
Access to the full range of evidence-based treatment options, including medications, topical therapies, pulsed radiofrequency of the dorsal root ganglion, nerve blocks, and multidisciplinary support.
Specialist experience in interventional procedures including PRF of the DRG, which has moderate evidence in PHN and is not widely available outside specialist pain practice.
Management of PHN affecting the face, head, and eye area, including ophthalmic and trigeminal distribution cases.
A diagnosis-first approach: we do not offer treatment without understanding what is driving your pain.
Communication and coordinated care with your GP and other treating clinicians.
We see patients who have often waited a long time, tried multiple medications, and been told there is nothing more to do. In many cases, there is. The right starting point is an accurate assessment of what is driving the pain and a structured conversation about what the evidence supports.
What people say about living with post-herpetic neuralgia
In our clinics, and through patient communities and published online forums, we hear common patterns from people living with post-herpetic neuralgia. These experiences help shape how we listen and assess each new patient. Descriptions that come up regularly in these forums and discussions include:
A burning, stinging, or raw sensation that persists in the area where the shingles rash appeared, often long after the skin looks normal
Pain that is noticeably worse at night, disrupting sleep and contributing to fatigue
Extreme sensitivity to touch in the affected area: clothing, bedsheets, water from a shower, or a light breeze can feel intensely painful
Sharp, shooting, or electric shock sensations that come without warning
Numbness or an altered sensation in the skin alongside the pain
Pain that feels disproportionate to what can be seen: normal-looking skin that is acutely painful to any contact
Frustration at being told the shingles is over when the pain has not resolved, and in some cases has worsened
Frequently Asked Questions (FAQ)
1. How long does post-herpetic neuralgia last?
PHN is defined as pain lasts for three months or more after shingles onset, but duration varies considerably between individuals. Some patients experience gradual resolution over six to twelve months. Others have pain that persists for years.
Older age, more severe initial shingles pain, and delay in treatment are associated with longer duration. The absence of improvement with standard medication is not a reason to assume the condition is permanent. It is a reason to seek specialist assessment.
2. Can post-herpetic neuralgia be treated effectively?
Patients can achieve meaningful improvement with appropriate treatment. First-line medications help a proportion of patients. For those with inadequate response, targeted interventional treatments, particularly pulsed radiofrequency of the dorsal root ganglion, could lead to pain reduction, evidence has shown. The key is identifying which approach best matches the mechanism driving the pain in that individual.
No single treatment works for everyone, which is why specialist assessment is important before concluding that options have been exhausted.
3. Why does my shingles pain feel worse at night?
Night-time pain is one of the most commonly reported features of post-herpetic neuralgia, and it can be deeply disruptive to sleep.
During the day, your brain is constantly processing movement, touch, temperature, and sound. This background activity can partially mask pain signals. At night, when that input drops away, pain has less competition and can feel sharper or more prominent.
Hormone levels also play a role. Cortisol, which helps buffer inflammation, is naturally lower in the evening. For people with sensitised skin, even light contact from bedclothes or sheets can trigger discomfort.
This is a recognised feature of neuropathic pain, not a sign that the shingles rash is returning or that something new is wrong. If night-time pain is significantly affecting your sleep, it is worth raising with your treating clinician, as poor sleep can worsen pain sensitivity over time.
4. Why does clothing or light touch cause pain after shingles?
This symptom is called allodynia, pain in response to stimuli that are not normally painful. It reflects sensitisation of the damaged nerve fibres, which now respond to light mechanical input as if it were harmful.
Allodynia is one of the clearest signs of neuropathic pain and indicates that the nerve has been significantly altered by the shingles episode.
It is not a sign that the skin is damaged. The problem is in the nerve, not the tissue.
5. Why hasn't my gabapentin or pregabalin stopped the pain completely?
Gabapentinoids work by broadly reducing nerve excitability across the nervous system. In PHN, particularly in earlier stages, the primary driver of pain is often localised to a specific dorsal root ganglion damaged by the virus.
A systemic medication that acts across the whole nervous system may not deliver sufficient modulation at that specific site without much higher doses which then could produce significant side effects.
For patients with incomplete response to medication, targeted interventional options such as pulsed radiofrequency delivered directly to the relevant DRG may address the source of pain more precisely.
6. What is pulsed radiofrequency of the dorsal root ganglion, and how is it different from radiofrequency ablation?
Conventional radiofrequency ablation (RFA) uses heat to disrupt a nerve, reducing pain signals from that nerve. In PHN, where sensory nerves are involved, ablation is generally not appropriate. Permanently disrupting a sensory nerve can create other problems including numbness or worse pain.
Pulsed radiofrequency (PRF) is different. It delivers radiofrequency energy in short pulses rather than continuously, and the temperature remains low enough that nerve tissue is not damaged.
The mechanism is thought to involve modulation of how the DRG processes and transmits abnormal pain signals, rather than ablation of the nerve itself. This makes PRF of the DRG relatively safe and appropriate for neuropathic conditions affecting sensory nerve roots, including PHN. It is performed as a day procedure under fluoroscopic guidance.
Pulsed radiofrequency is generally considered a low-risk procedure, though as with any interventional treatment, risks include temporary discomfort at the injection site, bruising, and in rare cases infection, nerve irritation, or nerve damage.
7. When should I see a pain specialist rather than continuing with my GP?
A specialist assessment is appropriate if pain has persisted for three months or more, if medications have provided only partial relief or are not well tolerated, or if allodynia and significant sleep disruption are present.
GPs play an important role in managing PHN and in coordinating care, but the range of treatment options available in a specialist pain setting, including interventional procedures, access to high-concentration topical agents, and multidisciplinary pathways, is substantially broader.
Waiting to see whether pain resolves on its own is reasonable in the early weeks after shingles, but ongoing poor control beyond three months warrants specialist review.
8. Can post-herpetic neuralgia affect the face?
Yes. When shingles affects the trigeminal nerve, particularly the ophthalmic division supplying the forehead, scalp, and eye, PHN in the facial distribution is a well-recognised complication. It can also affect the ear and jaw area.
Ophthalmic distribution PHN is associated with some of the highest pain intensities reported in any PHN presentation. Assessment and treatment differ from thoracic PHN in some respects, and specialist pain assessment is particularly important given the anatomical complexity and the range of targeted treatment options relevant to this distribution.
If shingles affects the eye or the skin around the eye, this requires urgent assessment by an ophthalmologist, as ophthalmic zoster can threaten vision and is considered a medical emergency.
9. Is there a time limit on when treatment can help?
There is no absolute time limit, but the evidence is clearest for interventional treatments, particularly PRF of the DRG, in patients with a duration of under two years.
Earlier treatment is associated with better outcomes, partly because central sensitisation is less established in earlier presentations.
Patients with longer-standing PHN can still benefit from treatment, but the approach may need to be broader, incorporating both interventional and multidisciplinary components.
The practical message is that earlier specialist assessment is better, but later assessment is still worth pursuing.
10. Does shingles vaccination reduce the risk of post-herpetic neuralgia?
Yes. The Shingrix vaccine (recombinant zoster vaccine, RZV), which is available in Australia, has demonstrated high efficacy against both shingles and post-herpetic neuralgia in adults over 50.
Vaccination does not eliminate risk entirely but substantially reduces both the likelihood of shingles and, if shingles does occur, the severity and duration of pain.
For adults over 50, and particularly those over 60 or with immune compromise, vaccination is an important preventive option worth discussing with a GP.
References
1. Johnson RW, Rice ASC. Post-herpetic neuralgia. New England Journal of Medicine. 2014;371(16):1526-1533.
One of the most cited clinical reviews of PHN, covering epidemiology, pathophysiology, clinical features, and treatment.
https://pubmed.ncbi.nlm.nih.gov/25317872/
2. Dworkin RH, Johnson RW, Breuer J, et al. Recommendations for the management of herpes zoster. Clinical Infectious Diseases. 2007;44(Suppl 1):S1-26.
Comprehensive evidence-based guidelines covering acute zoster management and prevention of PHN.
https://pubmed.ncbi.nlm.nih.gov/17143845/
3. Aggarwal A, Suresh V, Gupta B, Sonthalia S. Post-herpetic Neuralgia: A Systematic Review of Current Interventional Pain Management Strategies. J Cutan Aesthet Surg. 2020 Oct-Dec;13(4):265-274.
Systematic review and meta-analysis confirming moderate evidence for pain reduction with PRF of DRG and intercostal nerves in PHN patients.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8061658/
4. Ke M, Yinghui F, Yi J, Xeuhua H, Xiaoming L, Zhijun C, et al. Efficacy of pulsed radiofrequency in the treatment of thoracic postherpetic neuralgia from the angulus costae: a randomized, double-blinded, controlled trial. Pain Physician. 2013;16:15–25
Randomised double-blind trial demonstrating superior outcomes with PRF of DRG compared to sham in PHN. Pain reduction was assessed for 6 months.
https://pubmed.ncbi.nlm.nih.gov/23340530/
5. Kim YH, Lee CJ, Lee SC, Huh J, Nahm FS, Kim HZ, Lee MK. Effect of pulsed radiofrequency for postherpetic neuralgia. Acta Anaesthesiol Scand. 2008 Sep;52(8):1140-3.
PRF lesioning of DRG showed significant pain relief compared with the conventional treatments in patients with intractable PHN.
https://pubmed.ncbi.nlm.nih.gov/18840116/
6. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurology. 2015;14(2):162-173.
The foundational pharmacotherapy evidence base for neuropathic pain, including PHN. This comprehensive review underpins and directs current prescribing guidelines.
https://pubmed.ncbi.nlm.nih.gov/25575710/
7. Tyring SK. Management of herpes zoster and postherpetic neuralgia. Journal of the American Academy of Dermatology. 2007;57(6 Suppl):S136-42.
Practical clinical guidance on acute and post-herpetic management including antiviral and analgesic approaches.
https://pubmed.ncbi.nlm.nih.gov/18021865/
8. Ding Y, Yao P, Li H, Han Z, Wang S, Hong T, Zhao G. CT-Guided Stellate Ganglion Pulsed Radiofrequency Stimulation for Facial and Upper Limb Postherpetic Neuralgia. Front Neurosci. 2019 Mar 8;13:170.
This randomised controlled trial concluded that stellate ganglion pulsed radiofrequency treatment of facial and upper limb PHN was safe and effective.
https://pubmed.ncbi.nlm.nih.gov/30906243/
9. Shingles (herpes zoster) vaccine.
Australian Government, Department of Health, Disability and Aging. Information about the shingles vaccine, who it is recommended for, and how and where to get vaccinated. If you're eligible, you can get the shingles vaccine for free under the National Immunisation Program.
https://www.health.gov.au/topics/immunisation/vaccines/shingles-herpes-zoster-immunisation-service
10. Australian and New Zealand College of Anaesthetists and Faculty of Pain Medicine. Acute Pain Management: Scientific Evidence, 5th edition. 2020.
The primary Australian clinical reference for acute and perioperative pain, including evidence on zoster and neuropathic pain management.
https://www.anzca.edu.au/safety-and-advocacy/advocacy/college-publications/acute-pain-management-scientific-evidence